Quick Facts
Formation & Origin
Dolomite is a calcium magnesium carbonate that forms one of the great puzzles in geology: the 'dolomite problem.' Despite being the most common magnesium-bearing sedimentary mineral, scientists have struggled for over 200 years to synthesize dolomite at surface temperatures in the laboratory. It forms easily in nature but refuses to crystallize under controlled conditions, suggesting that biological activity (microbial processes) may play a crucial role in its formation.
The Dolomite Mountains of northern Italy - one of the most spectacular mountain ranges in Europe - are composed primarily of ancient dolomite rock, and both the mountains and the mineral are named after French mineralogist Deodat de Dolomieu, who first described the mineral in 1791.
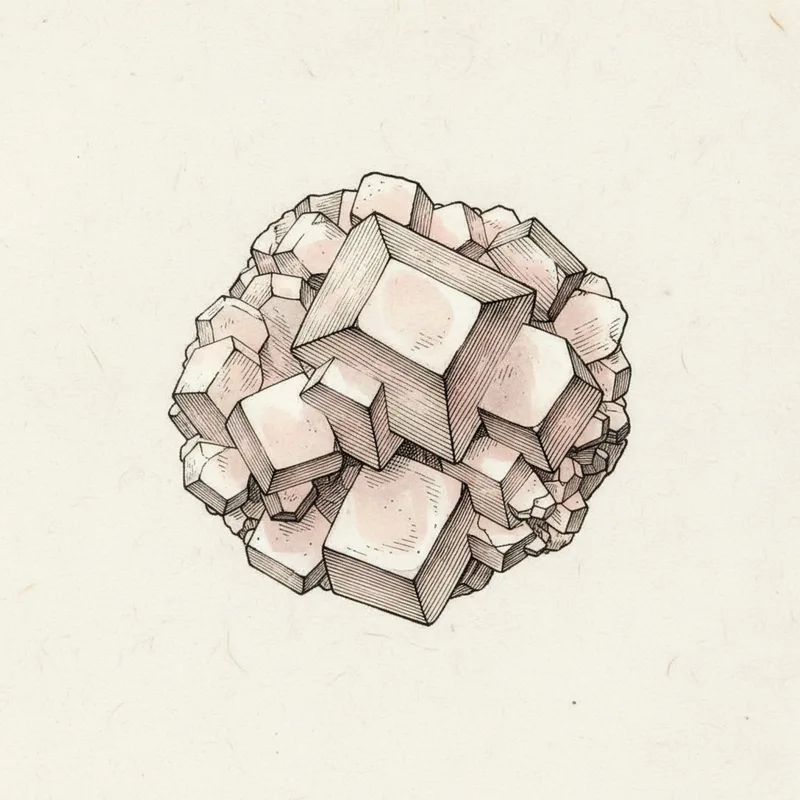
Dolomite crystals are famous for their distinctive curved 'saddle-shaped' or rhombohedral form. This curved crystal habit is almost unique to dolomite among common minerals and is one of its most recognizable features.
Identification Guide
Dolomite is identified by its curved saddle-shaped crystal habit, hardness of 3.5-4, and slow reaction with dilute hydrochloric acid (it fizzes only in hot acid or when powdered, unlike calcite which fizzes readily in cold dilute acid). This slow acid reaction is the key distinction from calcite.
Distinguish from calcite (stronger acid reaction, different crystal forms), magnesite (similar chemistry but rarely well-crystallized), and quartz (much harder, no acid reaction). The curved crystal faces visible on well-formed specimens are essentially diagnostic.
Spotting Fakes
Dolomite is common and affordable, so faking is unnecessary. The main identification challenge is distinguishing it from calcite, which looks similar. The acid test is definitive: a drop of cold dilute HCl on dolomite produces little or no fizzing, while calcite fizzes vigorously. Powdered dolomite will fizz in cold acid, so test on a crystal face, not a scratch.
Cultural & Metaphysical Traditions
Presented as cultural traditions, not scientific evidence
Dolomite is a gentle, quieter stone in the crystal healing world compared to more dramatic minerals. Practitioners associate it with centering, emotional stability, and reducing stress. Pink dolomite specifically is used for heart-centered calming work. The mineral's resistance to dissolving (the 'dolomite problem') is sometimes interpreted metaphysically as representing resilience and emotional stability.
Where It's Found
Named after these mountains, which are named after the mineral
Fine pink saddle-shaped crystals
Classic curved crystal specimens
Quality crystal specimens
Price Guide
Good to Know
Scratch test: At hardness 3.5, Dolomite can be scratched with a copper coin. Handle gently and keep away from harder stones in your collection.
Sources: Found in 4 notable locations worldwide, from Italy to Mexico.
Heft test: Dolomite has average mineral density (2.85). It feels about as heavy as you'd expect from a stone its size.
Explore More
Stay in the loop
From the Almanac
Updates from Crystal Almanac, when there’s something worth sharing.